Sample lifecycle
Accessioning with barcode verification. Aliquots, batches, retests, holds, rejections — every state change captured with who, when, why.
From the moment a sample is checked in to the moment a signed PDF lands with the clinic, FreshLIMS handles the workflow, the QC, the alerts and the paperwork. Built so you spend less time on the system and more time on the science.
Accessioning with barcode verification. Aliquots, batches, retests, holds, rejections — every state change captured with who, when, why.
Pull results in from analyzers, route through review, and feed back into reports. Configurable per-assay parsing.
Full Westgard rule set (1-3s, 2-2s, R-4s, 4-1s, 10x and 1-2s warning) on every QC point. One-click "open investigation" on a rejection.
Per-assay ranges by demographic, panic-value alerts that page the right people before the report is signed off.
Two-stage review with role-based approval. Each approval records the user, time and credential — printed on the PDF.
Every revision is preserved. Earlier versions stay accessible, addenda are flagged, retraction is one click.
Clinics submit orders directly into your queue. Sample status updates them live. PDF reports delivered automatically.
Pull billable orders into draft invoices, generate PDF invoices, record payments. The clinic sees the same invoice in their portal.
Append-only audit log of every meaningful action. Visitor logs, sign-in attempts, configuration changes — all captured.
Most labs lose months trying to validate a LIMS that wasn't built with the assessor in mind. We hand you the pack.
| What's in the pack | Why it matters |
|---|---|
| Software requirements specification mapped to ISO 15189 §6.7 (IT systems) and §7.x (process control) | Lets your QC manager show traceability between requirement, control, and feature. |
| IQ / OQ / PQ test scripts | Templates your team can execute and tick. No re-writing scripts from scratch. |
| Change control & release notes process | How you'll be told that v1.4 fixed a bug that affects your validated workflow. |
| Data integrity statement (ALCOA+) | Attributable, legible, contemporaneous, original, accurate. |
| Hosting / security / backup / DR white paper | SOC 2-lite description of what we run, where, encrypted how, backed up how often, restored how. |
| Cybersecurity posture | Encryption in transit + at rest, MFA, password policy, session timeouts, role-based access controls. |
| Data export / portability statement | Your lab's data is yours. Full export available on exit. |
Founding Lab tier customers receive the validation pack at sign-up. Lab Core and above include it as standard.
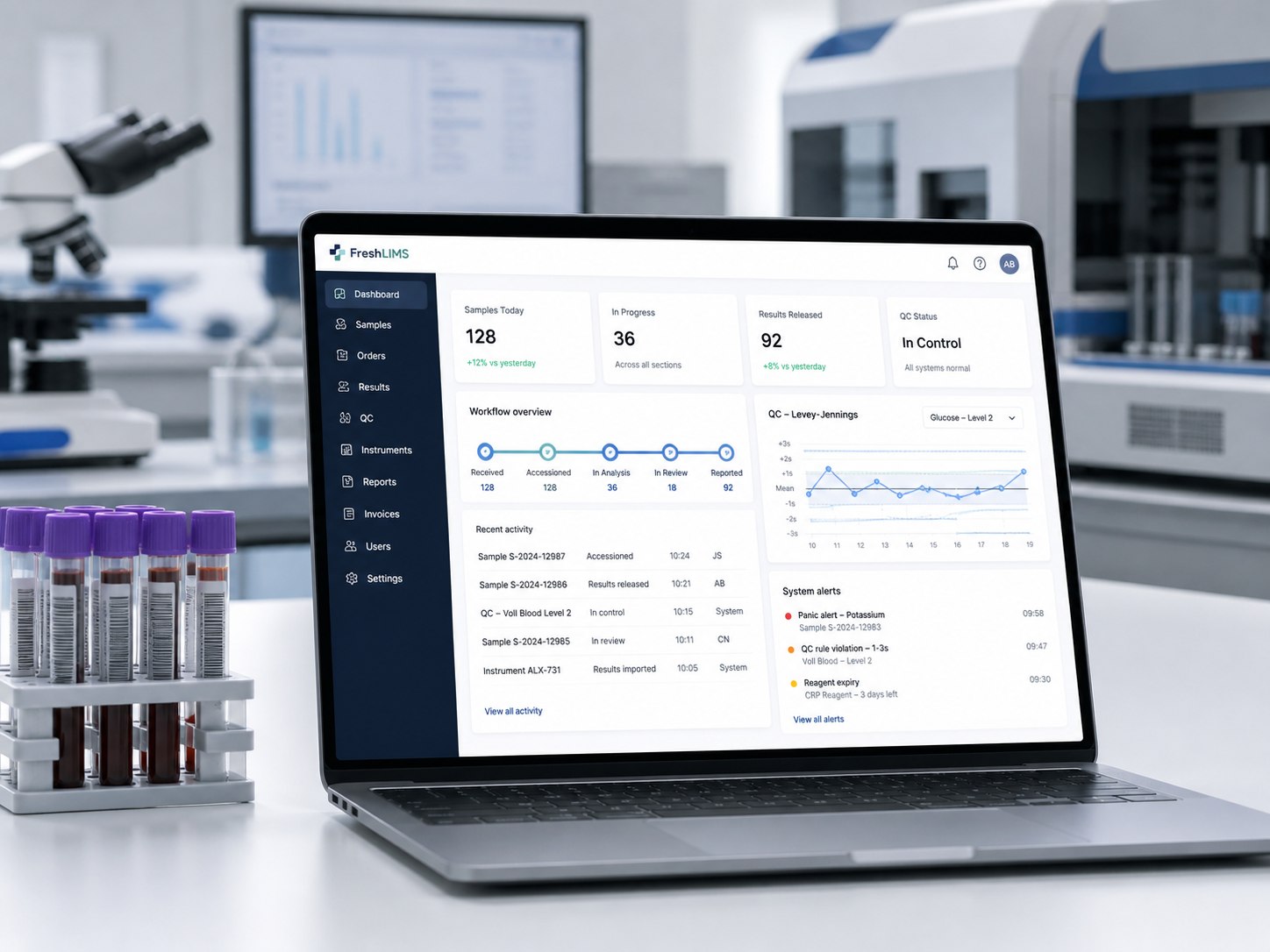
One of our customers — a UK clinical diagnostic lab — completed UKAS 15189 assessment and a CQC inspection running FreshLIMS as their LIMS. The validation pack covered the IT-systems clauses; their QC manager handed it to the assessor.
You get the same engine, the same pack, the same audit trail. We support the prep call when you need us.
Talk to sales